Collaborative Requirements Management
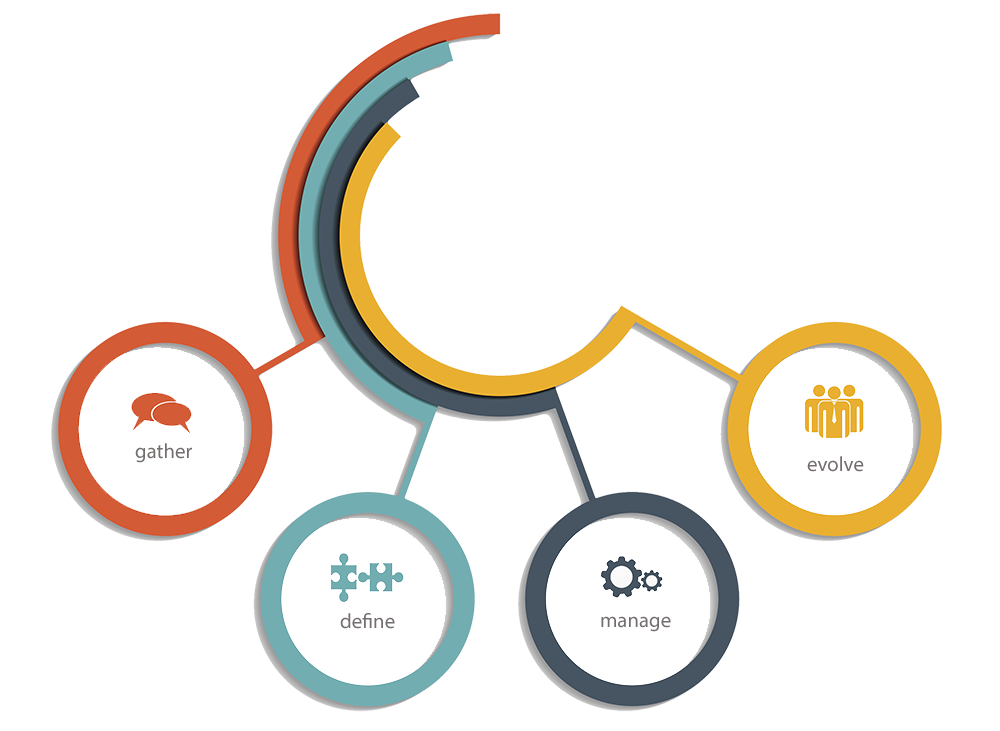
Gather, Define, Manage and Evolve are the underlying principles for DevSuite’s requirement management. Enterprises can take a more formal approach to requirements definition such as creating PRD’s, MRD’s, SRS, etc. whereas agile teams can define requirements in terms of user stories as part of the product backlog.
Collaborative Requirements Management Solution
Gather, Define, Manage and Evolve are the underlying principles for DevSuite’s requirement management. Enterprises can take a more formal approach to requirements definition such as creating PRD’s, MRD’s, SRS, etc. whereas agile teams can define requirements in terms of user stories as part of the product backlog.
Quantify Requirements to Drive Full Traceability
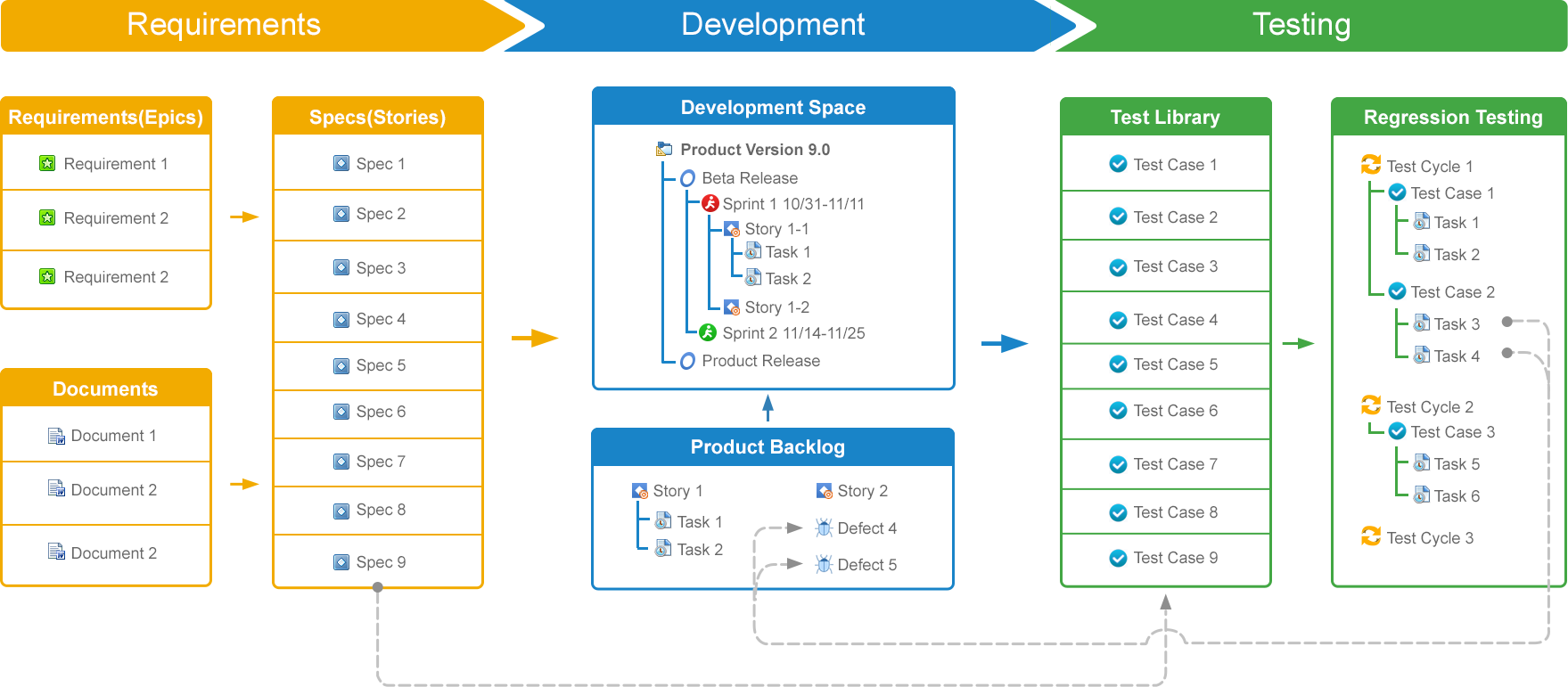
Trace back development tasks, defects, test cases and test tasks to requirements. Track requirement completion using development task rollups. Trigger flags to associated development tasks and test cases on any requirement change.
Baseline Requirements and Control Requirement Changes
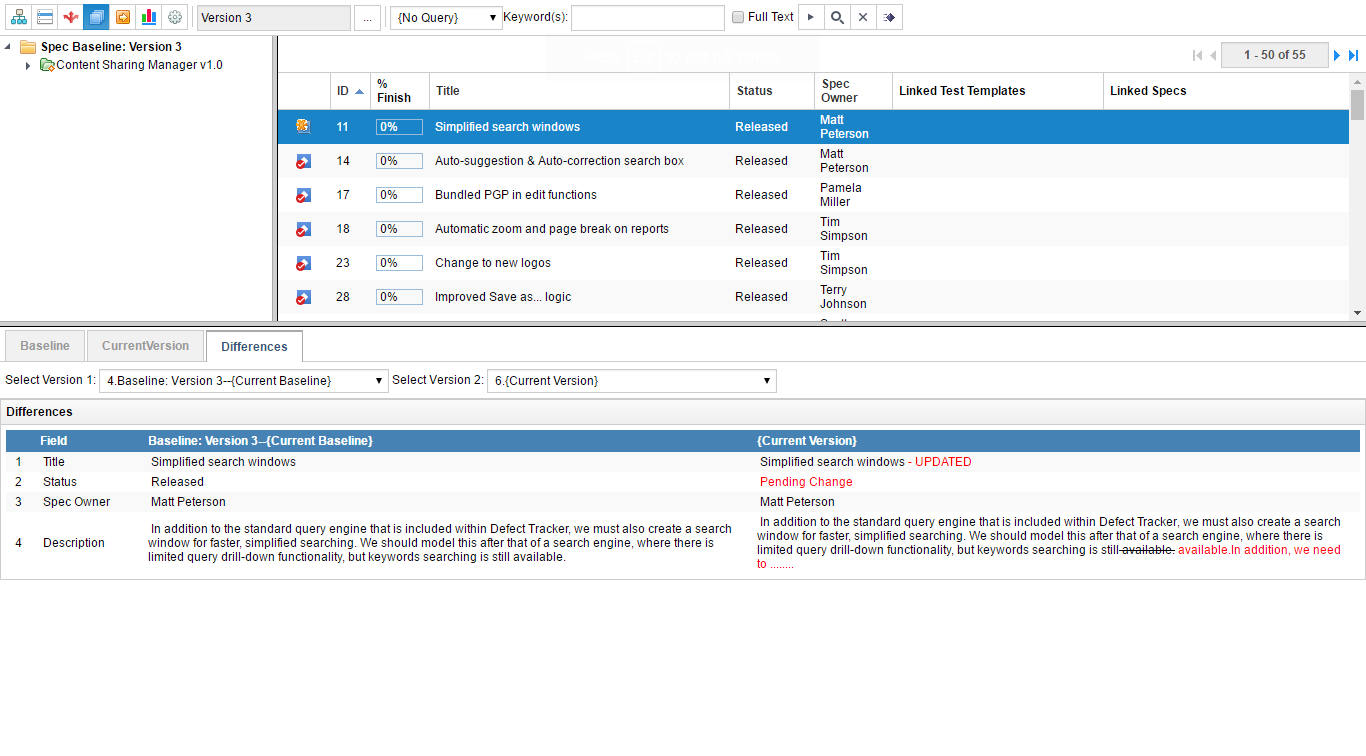
Baseline requirements once they have been finalized and approved. Use this baseline to track changes to a new version of the requirement and even rollback the changes to the baseline. Establish a change control procedure to approve any changes to the requirements.
Achieve Compliance
Normally achieving compliance with standards like IEC 62304 and FDA 21 CFR Part 11 and FDA General Principles of Software Validation would require sorting through mountains of documentation and acquiring costly point solutions. Our ALM saves you from all this by providing a simple solution to manage the entire lifecycle of your medical device software development.
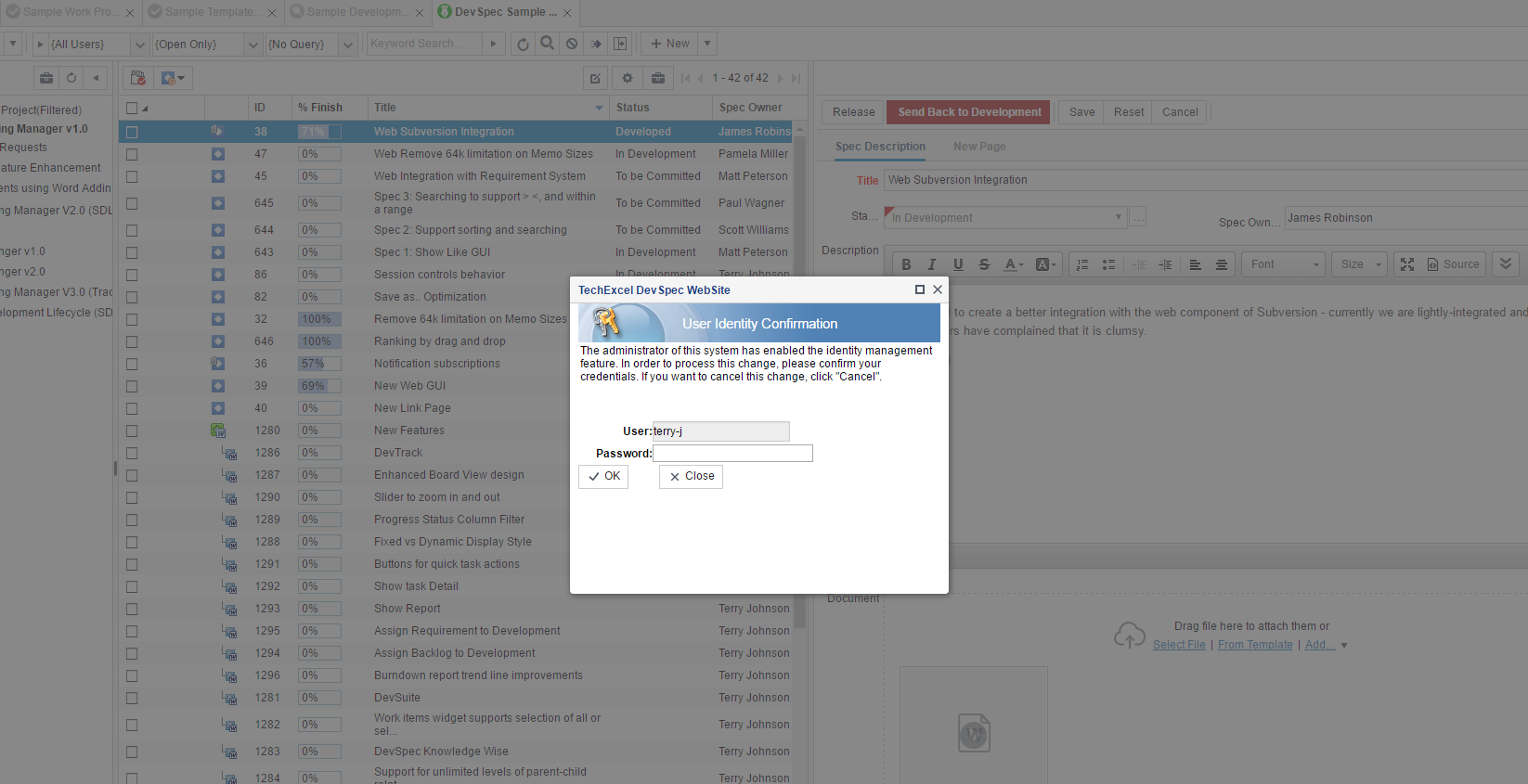
Seamless Integration with MS Word
Leverage MS Word’s editing capabilities with DevSpec’s powerful requirements management capabilities to create and manage requirements.
Key Features
Affordable Plans for Every Scenario
DevTrack with KnowledgeWise
- Agile Scrum or Kanban
- Simplified task or defect mgmt
- Single or multiple workflows
- Bulk or inline editing
- Collaborate with comments
- Full Audit Log
- Integrated with common KB
- Restful API available
DevTest with Jira Integration
- Test Case Mgmt & Execution
- Plan releases and cycles via wizards
- Submit defects directly into Jira or DevTrack
- Map fields from either system
- View test cases from defects
- Restful API available
DevSpec for Requirements
- Build requirements and specs
- Requirements traceability
- Roadmaps
- Baselines
- Unlimited relationship hiearchy
- MS Word Integration
- Full audit logs
- Custom Workflows
DevSuite for Total Traceability
- Single unified ALM platform
- Full traceability from requirements to testing
- Compliant with industry standards
- Quantifiable requirements
- Traditional or agile implementation
- Test case management and execution
- Built for scalability and performance
Questions?
Give Us a Call 1-800-439-7782
Customer Success Stories
Gael Quality was looking for a solution that could handle more than 15,000 test cases, multiple resources and the ability to fully integrate result from external sources.
Radon Labs’ main challenge was to find a solution that could easily handle more than 10,000 items per project, and TechExcel DevSuite was the only serious contender.
DevSuite resolves time zone differences and provides a collaborative environment for global software development teams.



